
We also provide extensive NCERT solutions, sample papers, NEET, JEE Mains, BITSAT previous year papers, which makes us a one-stop solution for all resources. 1995 - 2023.Physics Wallah is India's top online ed-tech platform that provides affordable and comprehensive learning experience to students of classes 6 to 12 and those preparing for JEE and NEET exams. Periodic Table of Elements - Calcium - Ca. If you need to cite this page, you can copy this text: This database focuses on the most common chemical compounds used in the home and industry. Molar mass calculations are explained and there is a JavaScript calculator to aid calculations. Molar Mass Calculations and Javascript Calculator.Introduces stoichiometry and explains the differences between molarity, molality and normality. Related ResourcesĪnswers many questions regarding the structure of atoms. Common Chemical Compounds of Calcium ReferencesĪ list of reference sources used to compile the data provided on our periodic table of elements can be found on the main periodic table page.Pure calcium is a shiny soft metal that will react violently with water to produce hydrogen. It is found in limestone (CaCO 3) gypsum (CaSO 4. Why did it take so long? Calcium is the fifth most abundant metalic element in the earth's crust, but is never found in the elemental form because it is so reactive. Davy was then successful in isolating the impure metal. Berzelius and Pontin prepared calcium amalgam by electrolizing lime in mercury. Additional Notes: Calcium was prepared as lime by the Romans under the name calyx in the 1st century A.D., but the metal was not discovered until 1808.Calcium is an essential component shells, bones, teeth and plant structures. Also used as an alloying agent for aluminum, copper and lead, a reducing agent for beryllium and used in fertilizer, concrete & plaster of paris. Uses of Calcium: Used for dehydrating oils, decarburization and desulfurization of iron and its alloys, getter in vacuum tubes. 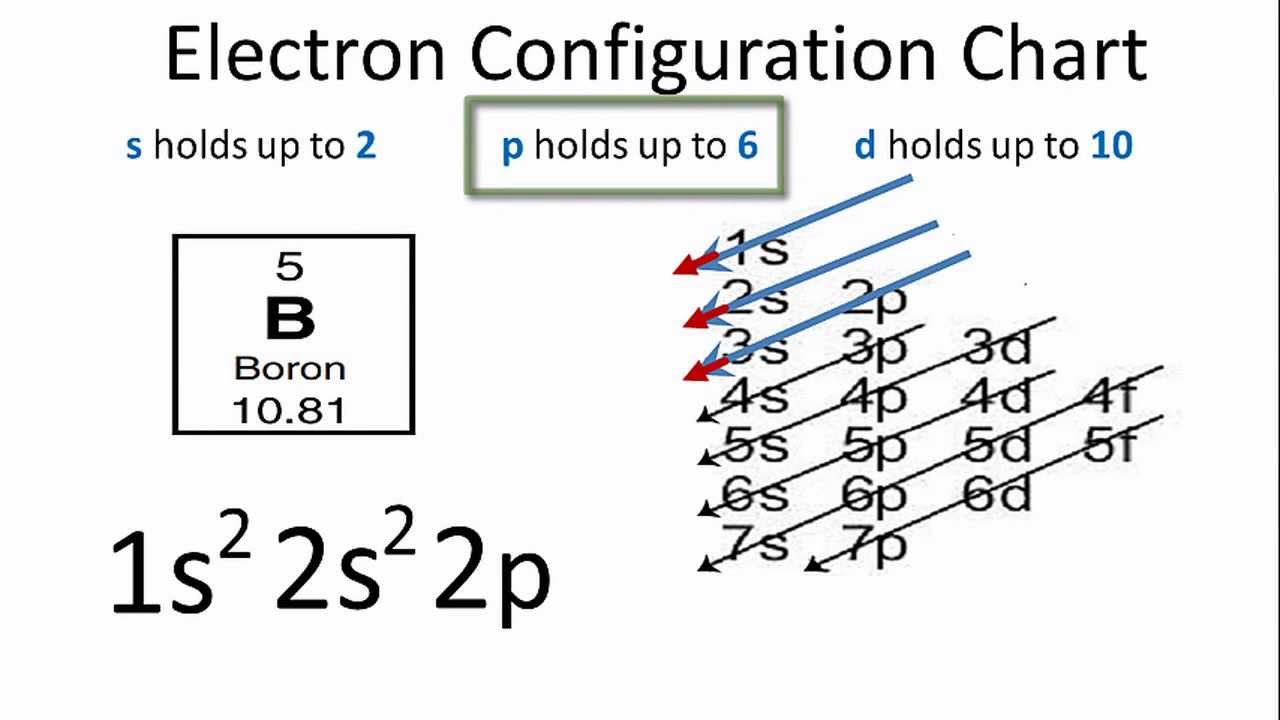
World production in 2000 was around 112,000,000 tons (CaO). Sources of Calcium: Obtained from minerals like chalk, limestone & marble.Name Origin: Latin: calx, calcis (lime).70kg human: 1 kg Who / Where / When / How Note: this data represents naturally occuring levels of elements in the typical human, it DOES NOT represent recommended daily allowances. Vapor Pressure = C Regulatory / Health.Enthalpy of Atomization: 184 kJ/mole 25☌.Description: silvery, soft metal, tarnishes to grayish white after exposure to air.Conductivity Electrical: 0.298 10 6/cm Ω.Coefficient of lineal thermal expansion/K -1: 22E -6.Valence Electron Potential (-eV): 29 Physical Properties of Calcium.Incompatibilities: water, oxidizers, acids, air, chlorine, chlorine tri-fluoride, fluorine, oxygen, silicon, sulfur.Electronegativity: 1 (Pauling) 1.04 (Allrod Rochow).Electrochemical Equivalent: 0.7477g/amp-hr.Valence Electrons: 4s 2 Electron Dot Model.Number of Neutrons (most common/stable nuclide): 20.Number of Electrons (with no charge): 20.Electrons per Energy Level: 2,8,8,2 Shell Model.Electron Configuration: 1s 2 2s 2p 6 3s 2p 6 4s 2.Crystal Structure: Cubic face centered.Cross Section (Thermal Neutron Capture)σ a/ barns : 0.43.Swedish: Kalcium Atomic Structure of Calcium.Series: Alkali Earth Metals Calcium's Name in Other Languages.Common Chemical Compounds of Calcium Overview of Calcium.In addition technical terms are linked to their definitions and the menu contains links to related articles that are a great aid in one's studies. Common chemical compounds are also provided for many elements. Skip to site menu on this page Periodic Table of Elements Element Calcium - CaĬomprehensive data on the chemical element Calcium is provided on this page including scores of properties, element names in many languages, most known nuclides of Calcium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
